

Friedreich’s ataxia (FRDA) is the most common hereditary ataxia and it´s an autosomal recessive degenerative disease.
The most common DNA abnormality associated with FRDA is the expansion of a GAA triplet repeat polymorphism localized in the first intron of the gene encoding frataxin (FXN). Depending on the number of repeats obtained, alleles can be classified into three different ranges:
Adellgene® Friedreich’s Ataxia is a semi-automated in vitro diagnostic kit designed for use in clinical laboratories which quantitatively determines the number of repetitions of GAA (guanine-adenine-adenine) in the first intron of the gene encoding frataxin (FXN) located in chromosome 9 resulting in Friedreich’s ataxia disease. It aims to aid the clinical diagnosis of Friedreich’s ataxia in individuals presenting compatible symptoms, that might span from mild to severe.
The kit allows the quantification of the size of normal, intermediate and expanded alleles with a size equal to or less than 200 repeats. The expansions of more than 200 repeats can be detected, but not quantified.
The procedure is based on the amplification by Triplet repeat Primed Polymerase Chain Reaction (TP-PCR) with fluorescent primers of genomic DNA extracted from whole blood and/or buccal swab, subsequent analysis of the amplified products in a capillary sequencer and number of repeats calculation from the corresponding amplified product size.
The patient referred by the corresponding health specialist (for example, neurologist) may be subjected to this determination and taking into account the compatibility of the symptoms presented. Among these patients there are, typically, neurological manifestations, such as progressive ataxia, dysarthria, decreased position sense and/or vibration sense in the lower limbs, weakness of the legs, extensor plantal responses; musculoskeletal manifestations, such as muscle weakness, scoliosis, hypertrophic non-obstructive cardiomyopathy; endocrinologic manifestations, such as diabetes mellitus; and/or optic atrophy or deafness. Besides these clinical manifestations, if there is family history.
The intended user of the kit is technical personnel trained to carry out the protocol and the interpretation of results described in the instructions for use.

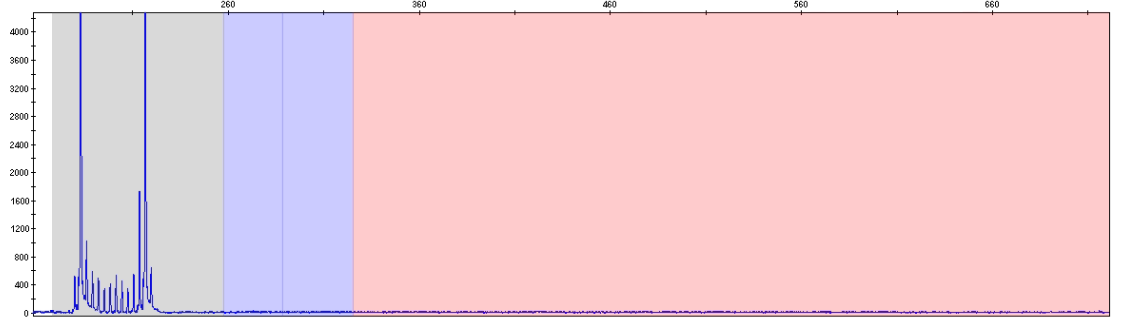
Sample with two wild-type alleles
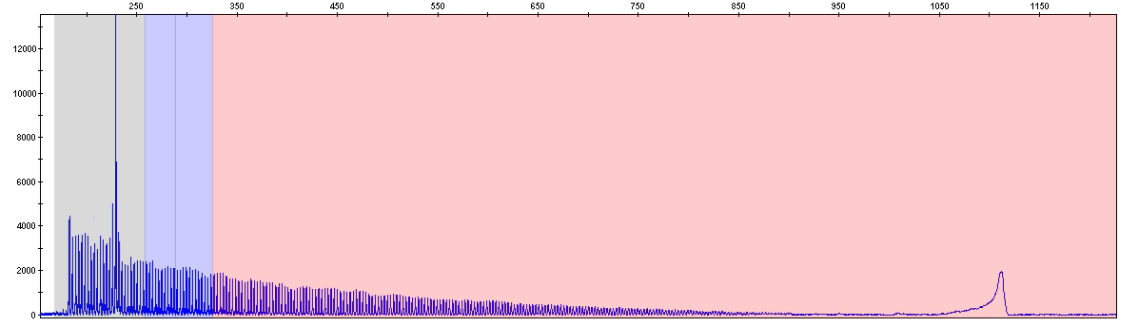
Sample with one normal allele and one expanded allele (>200 repeats)
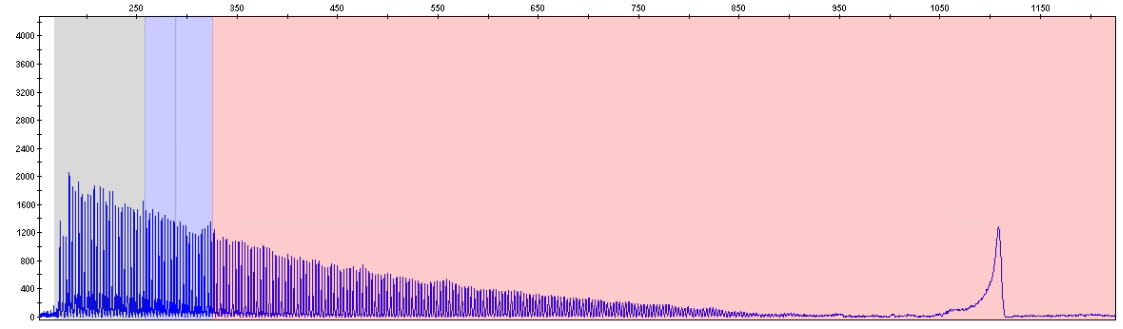
Sample with two expanded alleles (>200 repeats)
Log in to see the product files.




[Discontinued]
Kit for the determination of the CTG triplet repeats number of the DMPK gene by fluorescent fragment analysis

