

Idiopathic recurrent pregnancy loss (RPL) is a frequent obstetric complication, and an estimated 1-2% of women will suffer three or more miscarriages during their reproductive age, while almost 5% of women will suffer two or more RPLs. While the exact causes of RPL remain undetermined in most cases, genetic predisposition to venous thrombosis and elevation in total homocysteine (tHcy) levels (hyperhomocysteinemia) have been described as playing a role in the pathogenesis of RPL. Methylenetetrahydrofolate Reductase (MTHFR) Deficiency is the most common genetic cause of elevated levels of homocysteine in plasma.
The best-characterized MTHFR gene polymorphisms are the C677T and the A1298C missense single nucleotide polymorphisms. Individuals with both variants are found to have 50-60% wild-type MTFHR activity and, consequently, double heterozygosity for these two MTHFR polymorphisms is considered a risk factor for hyperhomocysteinemia.
Genvinset® MTHFR multiplex is a semi-automated kit for the in vitro qualitative detection of the A1298C polymorphism (NCBI dbSNP rs1801131; NM_001330358.2:c.1409A>C) and the C677T polymorphism (NCBI dbSNP rs1801133; NM_001330358.2:c.788C>T) in the Methylene tetrahydrofolate reductase (MTHFR) gene (OMIM: 607093) in genomic DNA extracted from whole blood using Real-Time PCR technology with specific TaqMan® probes.
The patient referred by the corresponding health specialist (reproductive clinician) and taking into account the compatibility of the symptoms presented; women suffering idiopathic recurrent pregnancy loss (RPL) may be subject to the determination of the polymorphism in the MTHFR gene. The results of this test should not be the only ones on which the therapeutic decision is based and should be used as an aid in the diagnosis together with results of other markers of the disease.
The intended user of the kit is technical personnel trained to carry out the protocol and the interpretation of results described in the instructions for use.

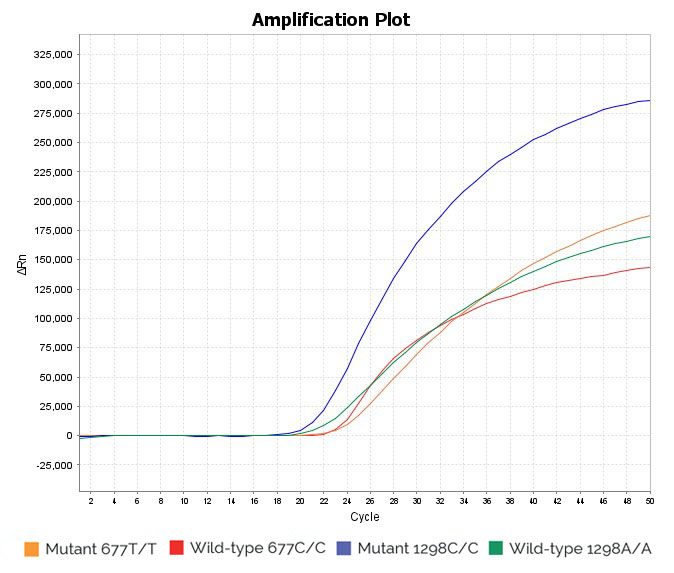
Heterozygous 1298A/C and 677C/T sample:
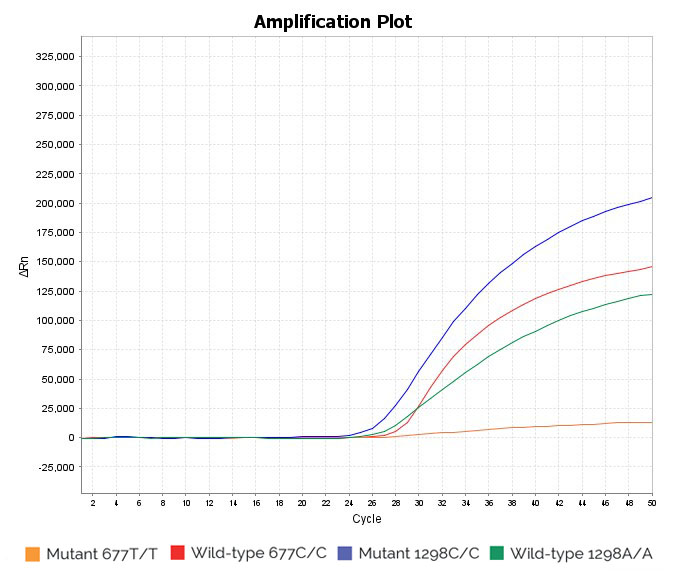
Heterozygous 1298A/C and homozygous wild-type 677C/C sample:
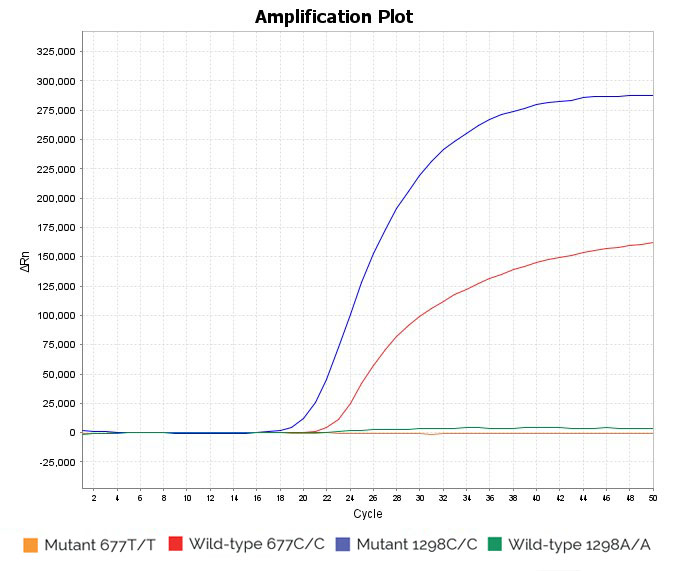
Homozygous mutant 1298C/C and homozygous wild-type 677C/C sample:
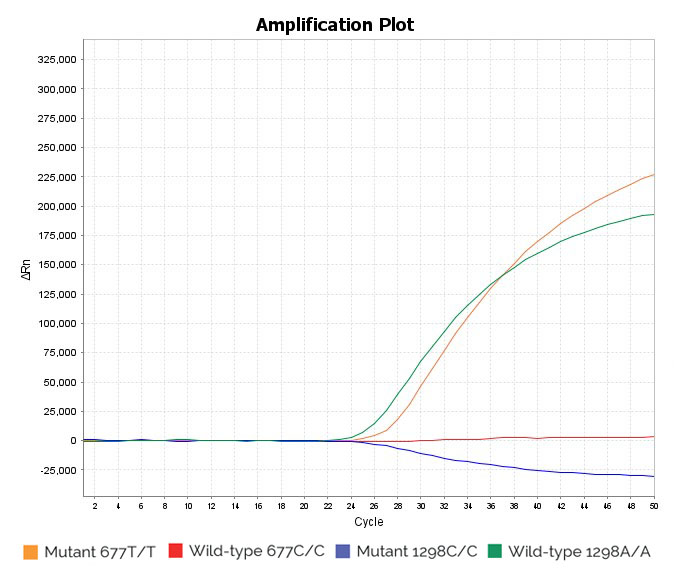
Homozygous wild-type 1298A/A and homozygous mutant 677T/T sample:
Log in to see the product files.













